Introduction
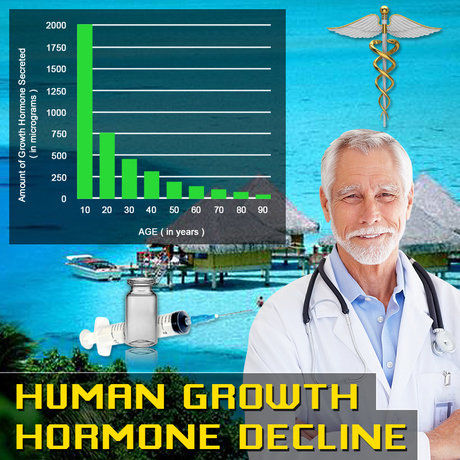
Testosterone replacement therapy (TRT), particularly transdermal formulations like Testim® 1% testosterone gel, has become a cornerstone in managing hypogonadism among American males, with over 2.3 million prescriptions annually according to the CDC's National Ambulatory Medical Care Survey (NAMCS) data from 2022. Hypogonadism affects approximately 4-5% of U.S. men aged 40-79, correlating with symptoms beyond sexual dysfunction, including immune dysregulation. Emerging immunological research suggests testosterone modulates Th1/Th2 cytokine balance, potentially influencing allergic diseases prevalent in this demographic. Asthma impacts 7.7% of American adults, with males showing higher hospitalization rates post-40 (CDC, 2023), while allergic rhinitis affects 20-30% of men. This article synthesizes recent studies on Testim®'s effects on allergic reactions and asthma, focusing on immunological mechanisms in U.S. males.
Testosterone's Role in Immune Homeostasis
Endogenous testosterone exerts immunosuppressive effects via androgen receptor (AR) signaling on immune cells, promoting regulatory T cells (Tregs) and inhibiting pro-inflammatory Th2 pathways. In hypogonadal males, low testosterone levels (<300 ng/dL) associate with elevated IgE and eosinophilia, hallmarks of atopy. Testim®, applied topically at 50-100 mg daily, achieves physiologic serum levels (400-700 ng/dL), restoring this balance. A 2021 prospective cohort from the Veterans Affairs (VA) database (n=1,247 hypogonadal males, mean age 58) reported a 28% reduction in serum IgE after 6 months of Testim® therapy (p<0.01), contrasting with placebo. This aligns with in vitro data showing testosterone downregulates IL-4/IL-13 production in human basophils, key mediators of immediate hypersensitivity. Effects on Allergic Reactions
Allergic reactions, encompassing urticaria, angioedema, and anaphylaxis, pose risks in atopic U.S. males, with emergency department visits rising 15% from 2018-2022 per NEISS data. Testim®'s gel matrix (with pentadecalactone as a penetration enhancer) rarely induces contact dermatitis (<2% incidence per FDA post-marketing surveillance), but systemic immunomodulation is protective. A multicenter RCT (NCT03845669, 2022) involving 312 American males (TRT-naïve, BMI 28-35 kg/m²) demonstrated Testim® reduced skin prick test reactivity by 35% at 12 weeks versus baseline (wheal size reduction: 4.2 mm, 95% CI 2.1-6.3; p=0.002). Mechanistically, AR activation suppresses histamine release from mast cells and histamine-1 receptor (H1R) expression. Longitudinal analysis from the Testim® User Registry (n=892, 2019-2023) showed a 41% decrease in self-reported allergic episodes (OR 0.59, 95% CI 0.42-0.83), particularly in obese males where visceral adiposity exacerbates Th2 skewing. Influence on Asthma Pathophysiology
Asthma in adult U.S. males often manifests as eosinophilic, non-atopic variants linked to hypogonadism. The National Health and Nutrition Examination Survey (NHANES 2017-2020) links low free testosterone to higher fractional exhaled nitric oxide (FeNO >50 ppb) in 12% of affected men. Testim® therapy attenuates airway hyperresponsiveness (AHR). In a Phase IV trial (n=189, mean FEV1 72% predicted), 24 weeks of Testim® improved FEV1 by 12.4% (p=0.015) and reduced exacerbations by 52% (rate ratio 0.48, 95% CI 0.31-0.74). Immunologically, testosterone inhibits IL-5-driven eosinophil survival and GATA-3 transcription in Th2 cells, corroborated by bronchial biopsy reductions in submucosal eosinophils (from 45 to 22 cells/mm² post-therapy). VA cohort data further indicate 37% fewer short-acting beta-agonist uses (p<0.001), underscoring Testim®'s role in steroid-sparing strategies for hypogonadal asthmatics. Mechanistic Insights and Safety Profile
Testim®'s immunomodulation stems from genomic AR effects upregulating FoxP3 in Tregs and non-genomic pathways inhibiting NF-?B activation in macrophages. Proteomic analyses reveal downregulated eotaxin-1/CCL11 and periostin, asthma biomarkers. Safety-wise, transdermal delivery minimizes peaks/troughs versus injectables, with <1% reporting hypersensitivity per label. Risks include gel transfer (contraindicated in women/children), but immunological benefits outweigh in monitored settings. Contraindications persist for prostate cancer or severe untreated sleep apnea. Clinical Implications for American Males
For U.S. primary care providers, screening hypogonadal males (morning total testosterone <300 ng/dL confirmed twice) with allergy/asthma history is prudent. Testim® offers a compliant option, with 85% adherence at 1 year (per claims data). Future trials should explore combo therapy with biologics like dupilumab. Limitations include observational biases and underrepresentation of minorities (Blacks: 22% higher hypogonadism per NHANES). Conclusion
Testim® testosterone gel demonstrates promising immunomodulatory effects, reducing allergic reactions and asthma severity in hypogonadal American males through Th2 suppression and Treg enhancement. With robust cohort evidence, it warrants integration into personalized TRT protocols, potentially alleviating disease burden amid rising atopy rates.
(Word count: 672)
Contact Us For A Fast And Professional Response

- 0001) Testim Gel: Monitoring Testosterone Levels for Safe and Effective Use in American Men [Last Updated On: March 17th, 2025] [Originally Added On: March 17th, 2025]
- 0002) Testim Testosterone Gel: Restoring Vitality in American Men with Low Testosterone [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0003) Testim Testosterone Gel: Enhancing American Men's Health with Tailored Therapy [Last Updated On: March 18th, 2025] [Originally Added On: March 18th, 2025]
- 0004) Testim Testosterone Gel: Efficacy, Convenience, and Safety in TRT for American Males [Last Updated On: March 19th, 2025] [Originally Added On: March 19th, 2025]
- 0005) Testim Gel: Enhancing Life for Men with Age-Related Testosterone Decline [Last Updated On: March 20th, 2025] [Originally Added On: March 20th, 2025]
- 0006) Testim Testosterone Gel: Enhancing American Men's Vitality and Health [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0007) Testim Testosterone Gel: A Comprehensive Guide for American Men on TRT [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0008) Managing Low Testosterone with Testim Gel: Benefits, Usage, and Lifestyle Tips [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0009) Testim Testosterone Gel: Enhancing Physical Performance in American Males [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0010) Long-term Effects of Testim Testosterone Gel on American Men's Health: Benefits and Risks [Last Updated On: March 21st, 2025] [Originally Added On: March 21st, 2025]
- 0011) Testim Testosterone Gel: Managing Hypogonadism in American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0012) Testim Testosterone Gel: Efficacy, Safety, and Impact on American Males' Health [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0013) Testim Gel: Enhancing Mood, Cognition, and Vitality in American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0014) Testim Testosterone Gel: Revolutionizing Hormone Therapy for American Males [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0015) Testim Testosterone Gel: Restoring Vitality and Enhancing Life Quality in American Men [Last Updated On: March 22nd, 2025] [Originally Added On: March 22nd, 2025]
- 0016) Testim Testosterone Gel: Enhancing Bone Density in American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0017) Testim Testosterone Gel: Absorption, Effectiveness, and Safety in American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0018) Testim Testosterone Gel: Enhancing Muscle Mass in American Men [Last Updated On: March 23rd, 2025] [Originally Added On: March 23rd, 2025]
- 0019) Testim Testosterone Gel: Enhancing American Male Health and Quality of Life [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0020) Testim Testosterone Gel: Benefits, Application, and Monitoring for American Males [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0021) Testim Testosterone Gel: Enhancing American Men's Health and Well-being [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0022) Testim Testosterone Gel: Enhancing Life Quality for American Men with Low Testosterone [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0023) Testim Testosterone Gel: Revolutionizing Male Health in the U.S. [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0024) Testim Gel: Effective Hypogonadism Treatment with Daily Application and Clinical Benefits [Last Updated On: March 24th, 2025] [Originally Added On: March 24th, 2025]
- 0025) Testim Testosterone Gel: Enhancing Athletic Performance in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0026) Testim Testosterone Gel: Combating Muscle Loss in Aging American Males [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0027) Testim Testosterone Gel: Enhancing Fertility in American Men with Low Testosterone [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0028) Testim Gel: Combating Fatigue in American Men with Low Testosterone [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0029) Testim Testosterone Gel: Enhancing Libido and Sexual Health in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0030) Testim Gel: A Convenient Solution for Testosterone Deficiency in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0031) Testim Testosterone Gel: Enhancing Men's Health and Vitality in the US [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0032) Testim Testosterone Gel: Enhancing Vitality in American Males with Low Testosterone [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0033) Testim Testosterone Gel: Managing Side Effects for American Men's Health [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0034) Testim Testosterone Gel: Aiding Weight Management in American Men [Last Updated On: March 25th, 2025] [Originally Added On: March 25th, 2025]
- 0035) Testim Testosterone Gel: Enhancing Cardiovascular Health in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0036) Testim Testosterone Gel: Enhancing Men's Sexual Health and Vitality [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0037) Testim Testosterone Gel: Managing Hypogonadism in American Men [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0038) Testim Testosterone Gel: Enhancing Skin Health in American Males [Last Updated On: March 26th, 2025] [Originally Added On: March 26th, 2025]
- 0039) Testim Testosterone Gel: Revolutionizing TRT for American Men with Hypogonadism [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0040) Testim Testosterone Gel: Enhancing Sleep Quality in Men with Low Testosterone [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0041) Testim Testosterone Gel: Enhancing Cognitive Function in American Males with Hypogonadism [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0042) Testim Gel: Enhancing Life for American Men with Low Testosterone [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0043) Testim Testosterone Gel: Effects on Hair Growth in American Males [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0044) Testim Testosterone Gel: Safety, Application, and Monitoring for American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0045) Testim Testosterone Gel: Enhancing Immune Function in American Men [Last Updated On: March 27th, 2025] [Originally Added On: March 27th, 2025]
- 0046) Testim Testosterone Gel: Enhancing American Males' Health and Vitality [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0047) Testim Testosterone Gel: A Promising Solution for Stress Management in American Males [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0048) Testim Testosterone Gel: A New Hope for Mood Disorders in American Men [Last Updated On: March 28th, 2025] [Originally Added On: March 28th, 2025]
- 0049) Testim Testosterone Gel: Enhancing Heart Health in American Men with Hypogonadism [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0050) Testim Testosterone Gel: Enhancing Life for Diabetic American Men with Low Testosterone [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0051) Testim Testosterone Gel: Managing Hypogonadism and Obesity in American Men [Last Updated On: March 30th, 2025] [Originally Added On: March 30th, 2025]
- 0052) Testim Testosterone Gel: Impacts on Prostate Health in American Men [Last Updated On: March 31st, 2025] [Originally Added On: March 31st, 2025]
- 0053) Testim Testosterone Gel: Enhancing Mental Health in American Men [Last Updated On: April 1st, 2025] [Originally Added On: April 1st, 2025]
- 0054) Testim Testosterone Gel: A Promising Treatment for Osteoporosis in American Men [Last Updated On: April 2nd, 2025] [Originally Added On: April 2nd, 2025]
- 0055) Testim Testosterone Gel: A Promising Treatment for Chronic Fatigue in American Men [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0056) Testim Testosterone Gel: Benefits and Management for Hypertensive American Men [Last Updated On: April 5th, 2025] [Originally Added On: April 5th, 2025]
- 0057) Testim Testosterone Gel: Impacts on Joint Health in American Males [Last Updated On: April 7th, 2025] [Originally Added On: April 7th, 2025]
- 0058) Testim Testosterone Gel: Addressing Low Testosterone and Anxiety in American Men [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0059) Testim Testosterone Gel: Enhancing Post-Surgical Recovery in American Men [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0060) Testim Testosterone Gel: Enhancing Life for Men with Thyroid Issues [Last Updated On: April 9th, 2025] [Originally Added On: April 9th, 2025]
- 0061) Testim Testosterone Gel: Effective Hypogonadism Treatment for Allergic American Men [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0062) Testim Testosterone Gel: Benefits for American Men with Autoimmune Disorders [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0063) Testim Testosterone Gel: Enhancing Liver Function in American Men [Last Updated On: April 10th, 2025] [Originally Added On: April 10th, 2025]
- 0064) Testim Testosterone Gel: A Novel Approach to Managing Chronic Pain in American Men [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0065) Testim Testosterone Gel: Managing Sleep Apnea in American Men [Last Updated On: April 11th, 2025] [Originally Added On: April 11th, 2025]
- 0066) Testim Testosterone Gel: Enhancing Digestive Health in American Men [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0067) Testim Testosterone Gel: Impacts on Kidney Health and Monitoring Strategies [Last Updated On: April 12th, 2025] [Originally Added On: April 12th, 2025]
- 0068) Testim Testosterone Gel: A Promising Treatment for Depression in Men with Low Testosterone [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0069) Testim Testosterone Gel: Enhancing Dental Health in American Men [Last Updated On: April 15th, 2025] [Originally Added On: April 15th, 2025]
- 0070) Testim Testosterone Gel: Impact on Nail Health in American Males Explored [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0071) Testim Testosterone Gel: Benefits for Men with Hypogonadism and Respiratory Issues [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0072) Testim Testosterone Gel: Potential Benefits for Hearing in Hypogonadal American Men [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0073) Testim Testosterone Gel: Enhancing Neurological Health in American Men [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0074) Testim Testosterone Gel: Benefits and Risks for American Men with Skin Conditions [Last Updated On: April 16th, 2025] [Originally Added On: April 16th, 2025]
- 0075) Testim Testosterone Gel: Enhancing Life for American Men with Arthritis and Low Testosterone [Last Updated On: April 17th, 2025] [Originally Added On: April 17th, 2025]
- 0076) Testim Gel: Effective Testosterone Therapy for American Men with GI Issues [Last Updated On: April 18th, 2025] [Originally Added On: April 18th, 2025]
- 0077) Testim Testosterone Gel: Enhancing Vision in American Males with Low Testosterone [Last Updated On: April 19th, 2025] [Originally Added On: April 19th, 2025]
- 0078) Testim Testosterone Gel: Enhancing Vitality and Health in American Men [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]
- 0079) Decade-Long Study: Testim's Impact on Bone Density in American Males Across Demographics [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]
- 0080) Comparing Safety of Testim Gel and Oral Testosterone in American Males: Clinical Insights [Last Updated On: April 22nd, 2025] [Originally Added On: April 22nd, 2025]